 It is critical to ensure that the problems found during the audit were addressed and respective action is taken to resolve them. Corrective action and Preventive action (CAPA) are an integral part of the audit process.įollow-up is an essential part of a successful audit. If in the audit any non-conformities or issues are identified, it is crucial to take action to resolve them and ensure that they are not repeated in the future. The auditor should provide a written report summarizing the audit results and any proposed corrective action. Post-audit, the auditor will meet with the stakeholders to discuss the findings and also provide any potential improvements that could be made. 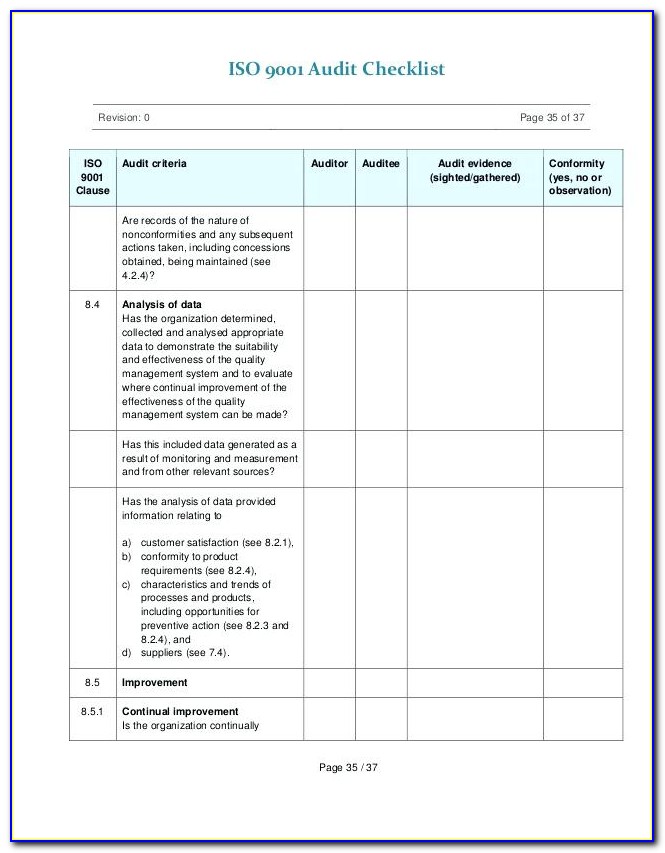
This depends totally on the organization and management.Īfter deciding when the audit is to be conducted, a concrete plan is required to ensure the availability of all the stakeholders on the scheduled date and time.Īn audit may consist of reviewing past records, interviewing employees, or observing the process itself. Internal audits are typically conducted twice a year or annually. This step must clearly define what is to be audited, who is responsible for the audit, and when the audit should be done. We have broken down the requirements for better understanding:Īn organization’s internal audit program must include documentation and all implementation components. The internal audit section in 13485 mentions that the organization performs an internal audit program, solves problems identified during the audit, and verifies if problems are resolved. Under section 8.2.4 'Internal audit,' firms are required to maintain a planned and documented arrangement for internal auditing and ensure there is no delay when corrective action is necessary. Any medical device manufacturing company is required to carry out the ISO 13485 internal audits regularly to ensure the quality and safety of the device. If you would like further information on our wide range of templates or have any suggestions for improvements that require support in addition to our templates in the form of other Medical Device Quality Assurance or Regulatory Affairs services, contact us at. This program should take into consideration the associated risk and importance of the processes being audited, as well as the outcomes of previous audits if any.The standard also requires that your organisation plans an audit program.any applicable regulatory requirements.any applicable requirements established by the organisation,.You must use these audits to ensure that the Quality Management System conforms to:.The standard requires that your organisation perform regular internal audits at planned intervals. 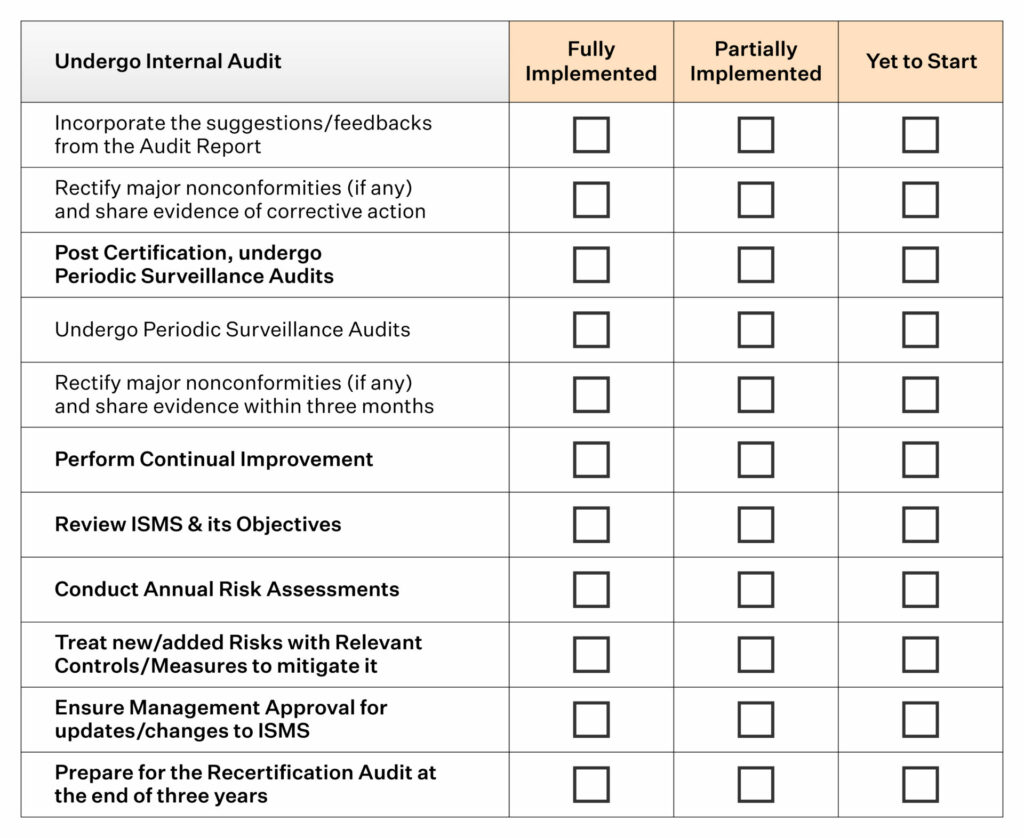
What does the standard say about Internal Audits? You will also set the objective(s) for the audit and reference the relevant section(s) of regulation that you will be assessing your QMS against.ISO 13485: The Essential Audit Checklist. The purpose of the Internal Audit Plan is to help your organisation set a day plan when performing internal quality audits. The Essential Checklist to an ISO 13485 Audit Free XLS Template - Dot Compliance.  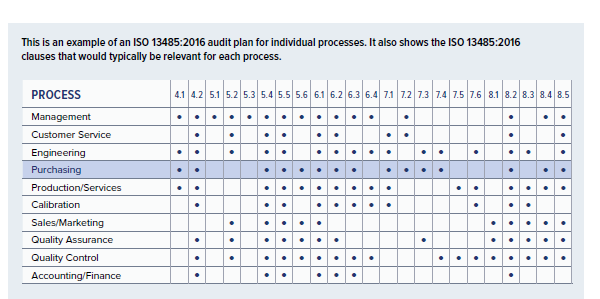
How will having an Internal Audit Plan & Checklist help my organisation? Your organisation will have to schedule regular assessments by both internal and external auditors.It covers sample policy for all process areas, Quality policy and organization structure and covers 1st tier of. Quality Manual: It covers sample copy of quality manual and requirement wise details for how ISO 13485:2016 is implemented. The assigned auditor will review your QMS documents, records and procedures and check your system for compliance with the standard. All the documents are related to ISO 13485:2016 for and user can edit it in line with their own processes.An audit is an independent examination of your Quality Management System (QMS) to ensure that it meets the requirements of ISO 13485.All the templates are fully editable Microsoft word documents allowing you to have complete control over the templates which can then be modified to suit your company branding/marketing requirements. We have carefully curated the templates to ensure they are easy to follow and adapt to make them specifically modified to your business needs. The templates have been specially written to ensure that they meet the basic requirements needed for each clause/section of the standard or regulation the templates relate to. Our Medical Device Quality Assurance and Regulatory Affairs document templates have been created by fully qualified and experienced professionals at Patient Guard, a Quality Assurance and Regulatory Affairs Service provider. QMS.8.2.4.2 - QR - Internal Audit Plan & Checklist Template 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
